THE SCIENCE BEHIND COOKING THAT TURKEY!
As many begin thinking about cooking their Thanksgiving meal, we are sharing this blog post as a look into the science behind cooking your turkey!

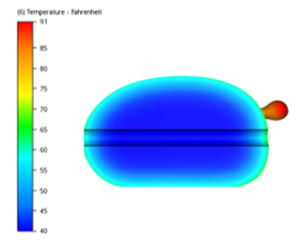
We begin this analysis by identifying the thermal conductivity of the bird. Per THERMOPHYSICAL PROPERTIES OF PROCESSED MEAT AND POULTRY
PRODUCTS, poultry’s thermal conductivity tracks linearly from 0.26 W/m-K to 0.48 W/m-K at room temperature to the final cook temperature of 175˚F. To put this in perspective the thermal conductivity of the aluminum pan on which the turkey is cooked is roughly 167 W/m-K. The poor poultry thermal conductivity is why the outer layer of the turkey will reach the fully cooked temperature, while the internal meat will still be cool. In larger birds, it is even more challenging to ensure the internal meat is cooked to a safe eating temperature. The other factor in the meat reaching the optimal internal temperature is the property of heat it will absorb per unit mass as it raises one Kelvin. (For reference 1K is equal to 1.8˚F) This property, called specific heat (Cp), is a constant determined by the internal molecular structure of the meat. For example; water has a specific heat of 4.187kJ/kg-K. This means for every kg of water it will absorb 4.187kJ of thermal energy as it raises one Kelvin. Again, per THERMOPHYSICAL PROPERTIES OF PROCESSED MEAT AND POULTRY PRODUCTS, the specific heat of poultry is 2.85 kJ/kg-K. This means that as the bird is conducting heat to its center it will also absorb 2.85 kJ/kg-K of thermal energy as it raises one Kelvin. This is why there is a temperature gradient across the bird during the cooking process, and also what determines the time it will take to cook the bird to the full 175˚F.
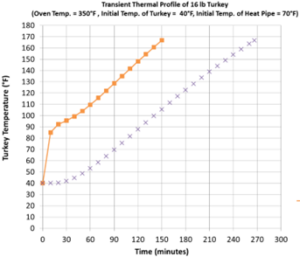
The bird; is made up of more than just the meat we eat. In a study conducted by Michele Marcotte and Ali R. Taherian published in THERMOPHYSICAL PROPERTIES OF PROCESSED MEAT AND POULTRY PRODUCTS, they identified that nearly 74% of a Turkey is moisture content with ~1.7% fat, and ~20% protein, salt, and carbohydrates. (These figures will vary between birds) This means that the thermal properties of cooking the bird will also be affected by the thermal properties of water. When a bird is frozen that 74% moisture content needs to thaw before the meat of the bird will be able to heat. The thawing process requires a significant amount of thermal energy to break the H2O crystal structure bonds. The energy required to melt a molecular structure is called the latent heat of fusion. Due to the hydrogen in the water molecule, water with a latent heat of fusion (344 kJ/kg) is higher than most other molecules. Thawing a turkey becomes particularly challenging because the moisture will hold a lot of thermal energy before it is able to transfer the heat inside the bird. This will give it a large temperature gradient and will cause the outer layer of meat to overcook, while the inner layer will be undercooked.

So, this Thanksgiving, make sure to give the bird ample time to conduct heat to the internal meat. And continuously cool down the outside of the turkey by basting the skin. And definitely wow your family with your superior thermal knowledge! Let them know that though the oven is really hot, the inside of the bird just won’t heat up that fast – poor thermal conductivity and all. Let them know that though it may seem like a good idea to put the frozen Turkey you forgot to thaw in the oven, the latent heat of fusion will work as a thermal dampener and absorb the energy you are trying to put in. This will create a greater temperature gradient and a significantly over cooked outer layer.
Happy Thanksgiving from Everyone at ACT!
Written by ACT’s very own Kimberly Mankosa
Lead Sales Engineer, Defense and Aerospace & Industrial Products
For more information on the thermal properties of your Thanksgiving Meal, check out this research article:
THERMOPHYSICAL PROPERTIES OF PROCESSED MEAT AND POULTRY PRODUCTS. Michèle Marcotte* (marcottem@agr.gc.ca) & Ali R. Taherian (taheriana@agr.gc.ca). Food Research and Development Centre, Agriculture and Agri-Food Canada, 3600 Casavant Blvd West. St. Hyacinthe, Quebec, J2S 8E3.
